Abstract— Despite the wide-ranging use of metal–organic frameworks (MOFs) as either electronic or ionic conductors, mixed electron–ion conductivity in MOFs remains largely unexplored. Here, we report a new methodology for designing new mixed ionic-electronic conductors (MIECs) by tethering ionophilic ethylene glycol (EG) functional groups onto an electrically conductive MOF (cMOF), yielding M3(HIR3-TAT)2 (M = Ni, Cu; TAT = triazatruxene; R = nBu, 1EG, 2EG; referred to as M-R). Leveraging systematic physicochemical variability in the pore microenvironment while preserving in-plane conjugation of the functionalizable TAT core, we orthogonally tune and enhance electronic conjugation through the crystalline framework (in-plane and out-of-plane) and ionic transport through defined abundant pore channels. Upon LiTFSI incorporation, Ni-1EG exhibits the highest room-temperature ionic conductivity (1.1 × 10–4 S/cm) in the series─nearly an order of magnitude higher than Ni-nBu (4.18 × 10–5 S/cm), which features a hydrophobic side chain of the same length─while preserving the electronic conductivity (∼5 × 10–4 S/cm). Consistent with previous reports, the added steric bulk in Ni-2EG reduces electronic conductivity to 2.7 × 10–4 S/cm, though the drop is minimal compared to aliphatic side chains of similar length. Counterintuitively, despite improved polarity, ionic conductivity significantly decreases in Ni-2EG, to 2.58 × 10–6 S/cm, likely due to pore blockage, with electronic conductivity largely remaining unchanged at 2.7 × 10–6 S/cm. This study unlocks the potential of cMOFs as tunable platforms for the decoupled control of ionic and electronic transport via side chain engineering. It opens doors to the unexplored design space of single-phase porous crystalline MIECs with orthogonal integration of functionalities tailored for emerging applications in chemistry, physics, and materials science.
Introduction— Mixed ionic-electronic conductive materials (MIECs) are of interest for a variety of applications across energy storage, electrocatalysis, memristors, chemiresistors, and bioelectronics. (1−7) In all of these applications, the general working mechanism requires conductive materials that respond electronically to ionic fluxes or vice versa. A major research challenge remains around achieving balanced ionic and electronic conduction within a single material, thereby eliminating the need for additives and composites. Conductive metal–organic frameworks (cMOFs), composed of π-conjugated ligands and metal ions forming van der Waals-stacked two-dimensional (2D) layers, have recently emerged as promising MIECs. Their long-range order enables high electronic conductivity via in-plane π-conjugation and out-of-plane orbital overlap along the framework (i.e., through-bond and through-space, respectively), while their abundant yet defined pore channels support efficient ionic transport. (8−13)
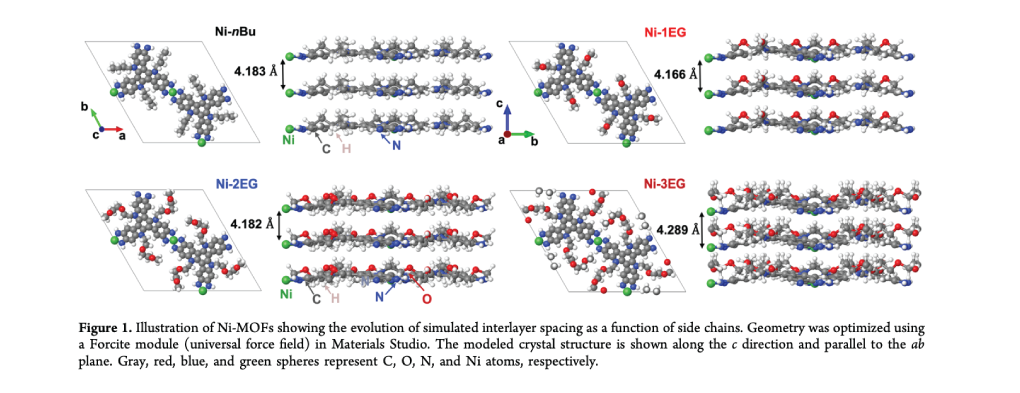
Unlike conventional semicrystalline or amorphous organic MIECs, where ionic conductivity is enhanced through bulk swelling, often disrupting the crystalline domains that underpin electronic conjugation, MOFs hold potential for intrinsic orthogonality of transport pathways. (10,11,14) That is, by leveraging ionic transport via side chain-modulated pore environment (e.g., polarity, pore size) and electronic transport through the framework (i.e., in-plane π-d conjugation and out-of-plane π–π stacking), the electronic conjugation can be largely preserved even upon tethering ionophilic side chains onto otherwise hydrophobic frameworks. With this property, cMOFs are promising in terms of overcoming the trade-off between the two modes of transport as well as their simultaneous optimization in a single material.
Despite their conceptual promise and wide-ranging use as either electronic or ionic conductors, mixed ionic-electronic conduction in MOFs has remained largely unexplored. Notably, a recent work has shown dual proton–electron conduction in single-phase three-dimensional (3D) MOFs via uncoordinated hydroquinone, highlighting the potential of functional group design for dual transport. (15) Furthermore, recent studies on triazatruxene (TAT)-based 2D cMOFs have shown that side chain functionalization with aliphatic groups of varying lengths can tune interlayer spacing by introducing steric hindrance while preserving in-plane conjugation. (16) In TAT MOFs, specific surface area and pore preoccupancy characterizations revealed that the side chains reside mainly within the pores. Notably, tuning of the interlayer spacing can be executed orthogonally to in-plane electronic conjugation. This strategy has been leveraged to precisely modulate spin concentration, Seebeck coefficients in thermoelectrics, and faradaic contributions in pseudocapacitors. (16−18)
These early successes encouraged us to explore synthetic approaches that allow installation of side chains, giving systematic insight into decoupling electronic and ionic conduction. Here, we present a molecular design strategy to enhance mixed conduction in cMOFs through side chain engineering, leveraging orthogonal control over ionic and electronic transport within an electrically conjugated, crystalline porous framework. Specifically, we employ ethylene glycol (EG) side chains, commonly used in physical mixtures with electrodes to enhance ion solvation and mobility while also mitigating interfacial incompatibility that hinders charge transfer at the electrolyte–electrode interface in solid-state electrochemical systems. (19) To our knowledge, this represents the first crystalline, side chain-functionalized cMOF deliberately designed as a mixed ionic-electronic conductor, in contrast to prior systems studied primarily as either ionic, electronic, or more recently protonic dual conductors.
 |
Aristide Gumyusenge
|
